THE CHALLENGE
The main challenge in developing effective gene and drug delivery technologies lies in achieving high efficiency without compromising cell health, which is critical for commercial and clinical success. Current methods like viral vectors pose safety risks and non-viral carriers often have low uptake, while physical techniques lack scalability for large or delicate cell populations. Electroporation, a popular physical method, offers promise due to its simplicity and broad applicability but suffers from inconsistent results, significant cell damage, and limited control over the delivery process. Additionally, standard testing methods fail to fully capture the extent of cellular stress and recovery, making it difficult to optimize protocols for sensitive cells such as stem cells. This creates a gap in the market for a reliable, scalable, and safe intracellular delivery platform that balances efficiency and cell viability while providing real-time insights to improve outcomes, especially for high-value therapeutic and research applications.
OUR SOLUTION
We offer a breakthrough platform that combines advanced nanofiber scaffolds with precise electrical stimulation inside a microfluidic device to improve gene and drug delivery into cells. By growing cells on specially designed polymer fibers that guide their shape and simultaneously measure their mechanical response in real time, we can finely control how electric pulses create temporary openings in the cell membrane while minimizing damage. This approach allows us to optimize delivery efficiency and cell survival, especially for delicate and rare primary cells, which are often difficult to treat with existing methods. The system’s ability to monitor cell behavior continuously without harmful dyes and adjust electric pulse parameters based on real-time feedback provides unmatched precision and scalability. This makes it a powerful tool for researchers and companies focused on developing safer and more effective therapies and cell engineering applications.
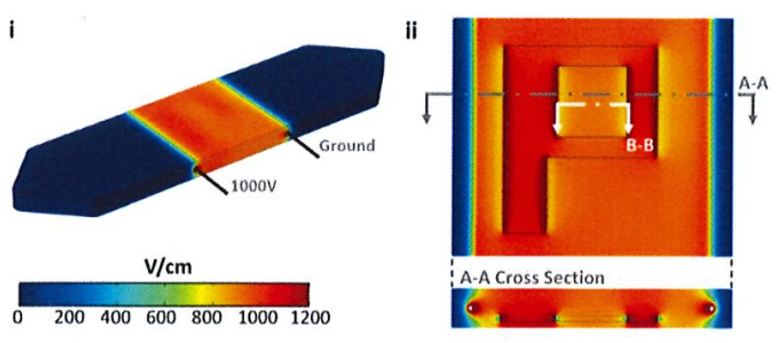
Figure: A finite element model shows the electric field distribution (i) along the walls of the fluidic channel and (ii) in the plane of the nanofiber scaffold.
Advantages:
- Over 60% transfection efficiency in rare primary T-cells
- Greater than 80% cell viability post-electroporation, reducing cell loss and costs
- Real-time, label-free mechanical monitoring for precise control and optimization
- Significant reduction in reagent uses and per-experiment costs through microfluidic integration and reusable scaffolds
Potential Application:
- Ex vivo manufacturing of engineered cell therapies like CAR-T and stem cells
- Gene editing and non-viral transfection for research and screening
- Development of stable producer cell lines for biopharmaceuticals
- High-throughput electroporation platforms with mechanobiology monitoring